|
9/20/2023 4 Comments Caesium isotopes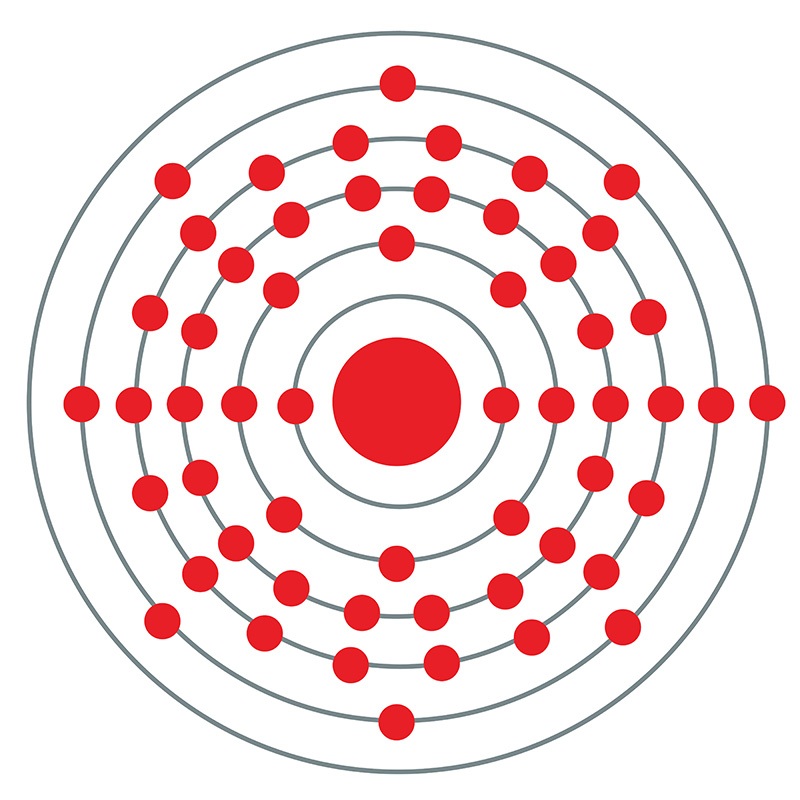
Reasonable agreement with the available experimental data. 137 Cs, as one of more than 30 isotopes of cesium, is fission a product of 235 U, whose decay chain is shown in Figure 1 3,4. The most important exposure routes are external exposure to the radiation emitted by the radioisotope and ingestion of radioactive cesium-contaminated food sources. 137 Cs (T½ 30.17 y) is among the most important hazardous radionuclides involved in radiological pollution to humans and the environment due to its long half-life and high fission yields 1,2. They can also be defined as atoms that contain an unstable combination of neutrons and protons, or excess energy in their nucleus. Radioisotopes are radioactive isotopes of an element. Mit einer Halbwertszeit von 30,17 Jahren zerfällt Cäsium-137 unter beta-Strahlung zu 94,6 in Barium-137m, das wiederum mit einer Halbwertszeit von 2,55 Minuten in das stabile Barium-Isotop Barium-137 zerstrahlt und für die Aussendung von Gammastrahlen verantwortlich ist das restliche Cäsium-137 zerfällt direkt zu 137 Ba. $I\sim\hbar\omega$ and energy spectra as well as $B(M1)/B(E2)$ values show Radioactive isotopes of cesium are a greater health concern than stable cesium. Different isotopes of the same element have the same number of protons in their atomic nuclei but differing numbers of neutrons. Isotopes, the possible chiral candidates and the evolution of three-dimensional Download a PDF of the paper titled The evolution of the chiral symmetry in cesium isotopes, by Duo Chen and 1 other authors Download PDF Abstract: Following the reports of candidate chiral doublet bands observed in cesium
4 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
